CAZypedia celebrates the life of Senior Curator Emeritus Harry Gilbert, a true giant in the field, who passed away in September 2025.
CAZypedia needs your help!
We have many unassigned pages in need of Authors and Responsible Curators. See a page that's out-of-date and just needs a touch-up? - You are also welcome to become a CAZypedian. Here's how.
Scientists at all career stages, including students, are welcome to contribute.
Learn more about CAZypedia's misson here and in this article. Totally new to the CAZy classification? Read this first.
Difference between revisions of "Syn/anti lateral protonation"
Harry Brumer (talk | contribs) m |
Wim Nerinckx (talk | contribs) |
||
| Line 8: | Line 8: | ||
== Overview == | == Overview == | ||
| − | |||
| − | == Table of syn/anti protonation examples == | + | |
| + | This page provides a table on the spatial positioning of the catalytic [[general acid]] residue in the active sites of [[glycoside hydrolase]]s. The table below updates those found in the seminal paper on this concept by Heightman and Vasella <cite>HeightmanVasella1999</cite>, and a following paper by Nerinckx ''et al.'' <cite>Nerinckx2005</cite>. | ||
| + | |||
| + | |||
| + | |||
| + | == Background == | ||
| + | |||
| + | |||
| + | |||
| + | The ''"not from above, but from the side"'' concept of semi-lateral glycosidic oxygen [[General_acid/base|protonation]] by [[glycoside hydrolase]]s was introduced by Heightman and Vasella <cite>HeightmanVasella1999</cite>. It was originally only described for [[Anomeric centre (alpha and beta)|beta]]-equatorial [[glycoside hydrolase]]s, but appears to be equally applicable to enzymes acting on an [[Anomeric centre (alpha and beta)|alpha]]-axial glycosidic bond <cite>Nerinckx2005</cite>. When dividing [[Sub-site nomenclature|subsite -1]] into half-spaces by the Ox, C1' and H1' plane of the –1 glycoside, many ligand-complexed structures show that the [[General_acid/base|proton donor]] is positioned either in the ''syn'' half-space, near the ring-oxygen of the –1 glycoside, or in the ''anti'' half-space at the opposite side of the ring-oxygen. Members of the same GH [[Families|family]] are always ''syn'' or always ''anti'' [[General_acid/base|protonators]] and this specificity appears to be preserved within [[Clans|clans]] of [[Families|families]]. This page's compilation of [[Sub-site nomenclature|subsite -1]] occupied complexes shows that about 70% of all GH [[Families|families]] are ''anti'' [[General_acid/base|protonators]]. | ||
| + | |||
| + | |||
| + | |||
| + | Closer inspection of [[Sub-site nomenclature|–1/+1 subsite]]-spanning ligands in crystal structures reveals a further intriguing corollary <cite>Nerinckx2005</cite>, <cite>Wu2012</cite>. In ''anti'' [[General_acid/base|protonating]] GH enzymes, the scissile [[Anomeric centre (alpha and beta)|anomeric bond]] (often as thio-analogue) always shows a dihedral angle φ (O5'-C1'-[O,S]x-Cx) that is in the lowest- energy synclinal (gauche) position. A minus synclinal dihedral angle φ for an equatorial glycosidic bond, or plus synclinal for an axial bond <cite>Perez1978</cite>, allows for hyperconjugative overlap of the C1'-O5' antibonding orbital with an antiperiplanar-oriented lone pair orbital lobe of the glycosidic oxygen, thereby creating partial double bond character and stabilization of the glycosidic bond by 4–5 kcal/mol; this ground-state stabilizing phenomenon is known as the ‘exo-anomeric effect’ <cite>Cramer1997</cite> <cite>Johnson2009</cite>. However, ''anti'' [[General_acid/base|protonation]] occurs precisely on the oxygen’s antiperiplanar lone pair, which removes the stabilizing effect, thus providing an additional enzyme-strategy for lowering the activation barrier en route to the [[Transition state|transition state]] (Figure 1 centre). | ||
| + | |||
| + | |||
| + | |||
| + | ''Syn'' [[General_acid/base|protonators]] show a different strategy <cite>Nerinckx2005</cite>, <cite>Wu2012</cite>. In many [[Sub-site nomenclature|–1/+1 subsite]]-spanning ligand complexes the dihedral angle φ of the scissile bond has been substantially twisted away from its lowest-energy synclinal position: clockwise to minus-anticlinal or antiperiplanar for beta-equatorial; counterclockwise to plus-anticlinal or antiperiplanar for alpha-axial [[Anomeric centre (alpha and beta)|anomeric bonds]]. This removes the hyperconjugative overlap and thus the stabilizing exo-anomeric effect. And because of this twisting, the lone pair of the glycosidic oxygen is pointing into the ''syn'' half- space, ready to be protonated by the ''syn''-positioned [[General_acid/base|proton donor]] (Figure 1 right). | ||
| + | |||
| + | |||
| + | |||
| + | [[File:Syn_anti.jpg|800px|thumb|center|Figure 1. Newman projections, with the glycosidic oxygen as proximal atom and the anomeric carbon as distal atom, showing ''anti'' (centre) versus ''syn'' (right) semi-lateral [[General_acid/base|protonation]] in beta-equatorial (top) and alpha-axial (bottom) [[glycoside hydrolase]]s. The indicated φ is the dihedral angle for O5'-C1'-O4-C4.]] | ||
| + | |||
| + | |||
| + | |||
| + | == Table of ''syn/anti'' protonation examples == | ||
| + | |||
=== Note === | === Note === | ||
| − | This table contains only one example per GH | + | |
| + | This table contains only one example per GH [[Families|family]] of a ligand-complexed protein structure where the ''syn'' positioning or ''anti'' positioning of the [[General_acid/base|proton donor]] can be clearly observed; other examples may be available on a [[Families|family-by-family]] basis. The reader is thus advised to consult the [http://www.cazy.org/fam/acc_GH.html#table CAZy database] for a current, comprehensive list of CAZyme structures. Where available, the selected examples are Michaelis-type complexes with the ligand spanning the [[Sub-site nomenclature|-1/+1 subsites]], since these have an intact glycosidic or thioglycosidic bond, or are ''N''-analogs of the substrate (''e.g.'' acarbose). In some examples, the [[General_acid/base|proton donor]] has been mutated (''e.g.'', to the corresponding amide or to an alanine), and in those cases one may wish to look at a superposition of the given PDB example with the structure of the native enzyme. If a Michaelis-type complex is not yet available, the second and third example choices, respectively, are trapped glycosyl-enzyme [[intermediate]]s and product complexes where [[Sub-site nomenclature|subsite -1]] is correctly occupied. | ||
| + | |||
| + | |||
''Please also be aware that this is a large table with many data, so some (hopefully few) errors may have sneaked in. Please contact the page Author or Responsible Curator with corrections.'' | ''Please also be aware that this is a large table with many data, so some (hopefully few) errors may have sneaked in. Please contact the page Author or Responsible Curator with corrections.'' | ||
| Line 1,035: | Line 1,063: | ||
# HeightmanVasella1999 Heightman, T.D. and Vasella, A.T. (1999) Recent Insights into Inhibition, Structure, and Mechanism of Configuration-Retaining Glycosidases. Angewandte Chemie-International Edition 38(6), 750-770. [http://www3.interscience.wiley.com/journal/55000581/abstract Article online]. | # HeightmanVasella1999 Heightman, T.D. and Vasella, A.T. (1999) Recent Insights into Inhibition, Structure, and Mechanism of Configuration-Retaining Glycosidases. Angewandte Chemie-International Edition 38(6), 750-770. [http://www3.interscience.wiley.com/journal/55000581/abstract Article online]. | ||
# Nerinckx2005 pmid=15642336 | # Nerinckx2005 pmid=15642336 | ||
| + | |||
| + | |||
| + | # Wu2012 pmid=23137336 | ||
| + | |||
| + | # Perez1978 Pérez S and Marchessault RH (1978) The exo-anomeric effect: experimental evidence from crystal structures. Carbohydr res 65, 114-120. | ||
| + | |||
| + | # Cramer1997 Cramer CJ, Truhlar DG and French AD (1997) Exo-anomeric effects on energies and geometries of different conformations of glucose and related systems in the gas phase and aqueous solution. Carbohydr res 298, 1-14. | ||
| + | |||
| + | # Johnson2009 pmid= 19733839 | ||
# Gloster2006 pmid=17002288 | # Gloster2006 pmid=17002288 | ||
# van_Bueren2009 pmid=18976664 | # van_Bueren2009 pmid=18976664 | ||
Revision as of 08:20, 14 November 2012
This page is currently under construction. This means that the Responsible Curator has deemed that the page's content is not quite up to CAZypedia's standards for full public consumption. All information should be considered to be under revision and may be subject to major changes.
- Author: ^^^Wim Nerinckx^^^
- Responsible Curator: ^^^Spencer Williams^^^
Overview
This page provides a table on the spatial positioning of the catalytic general acid residue in the active sites of glycoside hydrolases. The table below updates those found in the seminal paper on this concept by Heightman and Vasella [1], and a following paper by Nerinckx et al. [2].
Background
The "not from above, but from the side" concept of semi-lateral glycosidic oxygen protonation by glycoside hydrolases was introduced by Heightman and Vasella [1]. It was originally only described for beta-equatorial glycoside hydrolases, but appears to be equally applicable to enzymes acting on an alpha-axial glycosidic bond [2]. When dividing subsite -1 into half-spaces by the Ox, C1' and H1' plane of the –1 glycoside, many ligand-complexed structures show that the proton donor is positioned either in the syn half-space, near the ring-oxygen of the –1 glycoside, or in the anti half-space at the opposite side of the ring-oxygen. Members of the same GH family are always syn or always anti protonators and this specificity appears to be preserved within clans of families. This page's compilation of subsite -1 occupied complexes shows that about 70% of all GH families are anti protonators.
Closer inspection of –1/+1 subsite-spanning ligands in crystal structures reveals a further intriguing corollary [2], [3]. In anti protonating GH enzymes, the scissile anomeric bond (often as thio-analogue) always shows a dihedral angle φ (O5'-C1'-[O,S]x-Cx) that is in the lowest- energy synclinal (gauche) position. A minus synclinal dihedral angle φ for an equatorial glycosidic bond, or plus synclinal for an axial bond [4], allows for hyperconjugative overlap of the C1'-O5' antibonding orbital with an antiperiplanar-oriented lone pair orbital lobe of the glycosidic oxygen, thereby creating partial double bond character and stabilization of the glycosidic bond by 4–5 kcal/mol; this ground-state stabilizing phenomenon is known as the ‘exo-anomeric effect’ [5] [6]. However, anti protonation occurs precisely on the oxygen’s antiperiplanar lone pair, which removes the stabilizing effect, thus providing an additional enzyme-strategy for lowering the activation barrier en route to the transition state (Figure 1 centre).
Syn protonators show a different strategy [2], [3]. In many –1/+1 subsite-spanning ligand complexes the dihedral angle φ of the scissile bond has been substantially twisted away from its lowest-energy synclinal position: clockwise to minus-anticlinal or antiperiplanar for beta-equatorial; counterclockwise to plus-anticlinal or antiperiplanar for alpha-axial anomeric bonds. This removes the hyperconjugative overlap and thus the stabilizing exo-anomeric effect. And because of this twisting, the lone pair of the glycosidic oxygen is pointing into the syn half- space, ready to be protonated by the syn-positioned proton donor (Figure 1 right).
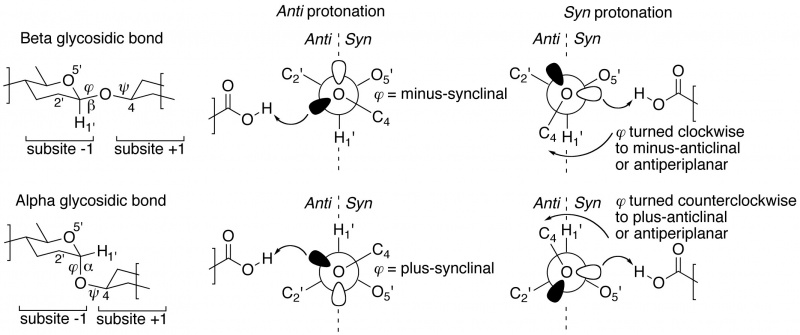
Table of syn/anti protonation examples
Note
This table contains only one example per GH family of a ligand-complexed protein structure where the syn positioning or anti positioning of the proton donor can be clearly observed; other examples may be available on a family-by-family basis. The reader is thus advised to consult the CAZy database for a current, comprehensive list of CAZyme structures. Where available, the selected examples are Michaelis-type complexes with the ligand spanning the -1/+1 subsites, since these have an intact glycosidic or thioglycosidic bond, or are N-analogs of the substrate (e.g. acarbose). In some examples, the proton donor has been mutated (e.g., to the corresponding amide or to an alanine), and in those cases one may wish to look at a superposition of the given PDB example with the structure of the native enzyme. If a Michaelis-type complex is not yet available, the second and third example choices, respectively, are trapped glycosyl-enzyme intermediates and product complexes where subsite -1 is correctly occupied.
Please also be aware that this is a large table with many data, so some (hopefully few) errors may have sneaked in. Please contact the page Author or Responsible Curator with corrections.
Table
This table can be re-sorted by clicking on the icons in the header (javascript must be turned on in your browser). To reset the page to be sorted by GH family, click the page tab at the very top of the page.
| Family | Clan | Structure fold | Anomeric specificity | Mechanism | Syn/anti protonator | Example PDB ID | Enzyme | Organism | Ligand | General acid | Nucleophile or General base | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| GH1 | A | (β/α)8 | beta | retaining | anti | 2cer | β-glycosidase S | Sulfolobus solfataricus P2 | phenethyl glucoimidazol | Glu206 | Glu387 | [7] |
| GH2 | A | (β/α)8 | beta | retaining | anti | 2vzu | exo-β-glucosaminidase | Amicolatopsis orientalis | PNP-β-d-glucosamine | Glu469 | Glu541 | [8] |
| GH3 | none | (β/α)8 | beta | retaining | anti | 1iex | exo-1,3-1,4-glucanase | Hordeum vulgare | thiocellobiose | Glu491 | Asp285 | [9] |
| GH5 | A | (β/α)8 | beta | retaining | anti | 1h2j | endo-β-1,4-glucanase | Bacillus agaradhaerens | 2',4'-DNP-2-F-cellobioside | Glu129 | Glu228 | [10] |
| GH6 | none | (β/α)8 | beta | inverting | syn | 1qjw | cellobiohydrolase 2 | Hypocrea jecorina | (Glc)2-S-(Glc)2 | Asp221 | debated | [11] |
| GH7 | B | β-jelly roll | beta | retaining | syn | 1ovw | endo-1,4-glucanase | Fusarium oxysporum | thio-(Glc)5 | Glu202 | Glu197 | [12] |
| GH8 | M | (α/α)6 | beta | inverting | anti | 1kwf | endo-1,4-glucanase | Clostridium thermocellum | cellopentaose | Glu95 | Asp278 | [13] |
| GH9 | none | (α/α)6 | beta | inverting | syn | 1rq5 | cellobiohydrolase | Clostridium thermocellum | cellotetraose | Glu795 | Asp383 | [14] |
| GH10 | A | (β/α)8 | beta | retaining | anti | 2d24 | β-1,4-xylanase | Streptomyces olivaceoviridis E-86 | xylopentaose | Glu128 | Glu236 | [15] |
| GH11 | C | β-jelly roll | beta | retaining | syn | 1bvv | xylanase | Bacillus circulans | Xyl-2-F-xylosyl | Glu172 | Glu78 | [16] |
| GH12 | C | β-jelly roll | beta | retaining | syn | 1w2u | endoglucanase | Humicola grisea | thiocellotetraose | Glu205 | Glu120 | [17] |
| GH13 | H | (β/α)8 | alpha | retaining | anti | 1cxk | β-cyclodextrin glucanotransferase | Bacillus circulans | maltononaose | Glu257 | Asp229 | [18] |
| GH14 | none | (β/α)8 | alpha | inverting | syn | 1itc | β-amylase | Bacillus cereus | maltopentaose | Glu172 | Glu367 | [19] |
| GH15 | L | (α/α)6 | alpha | inverting | syn | 1gah | glucoamylase | Aspergillus awamori | acarbose | Glu179 | Glu400 | [20] |
| GH16 | B | β-jelly roll | beta | retaining | syn | 1urx | β-agarase A | Zobellia galactanivorans | oligoagarose | Glu152 | Glu147 | [21] |
| GH17 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH18 | K | (β/α)8 | beta | retaining | anti | 1ffr | chitinase A | Serratia marcescens | (NAG)6 | Glu315 | internal | [22] |
| GH20 | K | (β/α)8 | beta | retaining | anti | 1c7s | chitobiase | Serratia marcescens | chitobiose | Glu540 | internal | [23] |
| GH22 | none | lysozyme type | beta | retaining | syn | 1h6m | lysozyme C | Gallus gallus | Chit-2-F-chitosyl | Glu35 | Asp52 | [24] |
| GH23 | none | lysozyme type | beta | inverting | syn | 1lsp | lysozyme G | Cygnus atratus | Bulgecin A | Glu73 | internal | [25] |
| GH24 | I | α + β | beta | inverting | syn | 148l | lysozyme E | Bacteriophage T4 | chitobiosyl | Glu11 | Glu26 | [26] |
| GH26 | A | (β/α)8 | beta | retaining | anti | 1gw1 | mannanase A | Cellvibrio japonicus | 2',4'-DNP-2-F-cellotrioside | Glu212 | Glu320 | [27] |
| GH27 | D | (β/α)8 | alpha | retaining | anti | 1ktc | α-N-acetyl galactosaminidase | Gallus gallus | NAGal | Asp201 | Asp410 | [28] |
| GH28 | N | β-helix | alpha | inverting | anti | 2uvf | exo-polygalacturonosidase | Yersinia enterocolitica ATCC9610D | digalacturonic acid | Asp402 | Asp381 Asp403 | [29] |
| GH29 | none | (β/α)8 | alpha | retaining | syn | 1hl9 | α-l-fucosidase | Thermotoga maritima | 2-F-fuco- pyranosyl | Glu266 | Asp224 | [30] |
| GH30 | A | (β/α)8 | beta | retaining | anti | 2v3d | glucocerebrosidase 1 | Homo sapiens | N-butyl-deoxynojirimycin | Glu235 | Glu340 | [31] |
| GH31 | D | (β/α)8 | alpha | retaining | anti | 2qmj | maltase-glucoamylase | Homo sapiens | acarbose | Asp542 | Asp443 | [32] |
| GH32 | J | 5-fold β-propeller | beta | retaining | anti | 2add | fructan β-(2,1)-fructosidase | Cichorium intybus | sucrose | Glu201 | Asp22 | [33] |
| GH33 | E | 6-fold β-propeller | alpha | retaining | anti | 1s0i | trans-sialidase | Trypanosoma cruzi | sialyl-lactose | Asp59 | Tyr342 | [34] |
| GH34 | E | 6-fold β-propeller | alpha | retaining | anti | 2bat | neuraminidase | Influenza A virus | sialic acid | Asp151 | Tyr406 | [35] |
| GH35 | A | (β/α)8 | beta | retaining | anti | 1xc6 | β-galactosidase | Penicillium sp. | d-galactose | Glu200 | Glu299 | [36] |
| GH37 | G | (α/α)6 | alpha | inverting | anti | 2jf4 | trehalase | Escherechia coli | validoxylamine | Asp312 | Glu496 | [37] |
| GH38 | none | (β/α)7 | alpha | retaining | anti | 1qwn | α-mannosidase II | Drosophila melanogaster | 5-F-β-l-gulosyl | Asp341 | Asp204 | [38] |
| GH39 | A | (β/α)8 | beta | retaining | anti | 1uhv | β-xylosidase | Thermoanaerobacterium saccharolyticum | 2-F-xylosyl | Glu160 | Glu277 | [39] |
| GH42 | A | (β/α)8 | beta | retaining | anti | 1kwk | β-galactosidase | Thermus thermophylus A4 | d-galactose | Glu141 | Glu312 | [40] |
| GH44 | none | (β/α)8 | beta | retaining | anti | 2eqd | endoglucanase | Clostridium thermocellum | cellooctaose | Glu186 | Glu359 | [41] |
| GH45 | none | 6-strand. β-barrel | beta | inverting | syn | 4eng | endo-1,4-glucanase | Humicola insolens | cellohexaose | Asp121 | Asp10 | [42] |
| GH46 | I | α + β | beta | inverting | predicted syn by clan | see at GH24 | ||||||
| GH47 | none | (α/α)7 | alpha | inverting | anti | 1x9d | α-mannosidase I | Homo sapiens | Me-2-S-(α-Man)-2-thio-α-Man | Asp463 | Glu599 | [43], [44] |
| GH48 | M | (α/α)6 | beta | inverting | predicted anti by clan | see at GH8 | ||||||
| GH49 | N | β-helix | alpha | inverting | predicted anti by clan | see at GH28 | ||||||
| GH50 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH51 | A | (β/α)8 | alpha | retaining | anti | 1qw9 | α-l-arabino- furanosidase | Geobacillus stearothermophilus | PNP-l-arabino-furanoside | Glu175 | Glu294 | [45] |
| GH53 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH54 | none | β-sandwich | alpha | retaining | anti | 1wd4 | α-l-arabino- furanosidase B | Aspergillus kawachii | l-arabinofuranose | Asp297 | Glu221 | [46] |
| GH55 | none | β-helix | beta | inverting | anti | 3eqo | β-1,3-glucanase | Phanerochaete chrysosporium K-3 | d-gluconolacton | Glu633 | unknown | [47] |
| GH56 | none | (β/α)7 | beta | retaining | anti | 1fcv | hyaluronidase | Apis mellifera | (hyaluron.)4 | Glu113 | internal | [48] |
| GH57 | none | (β/α)7 | alpha | retaining | anti | 1kly | glucanotransferase | Thermococcus litoralis | acarbose | Asp214 | Glu123 | [49] |
| GH59 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH63 | G | (α/α)6 | alpha | inverting | predicted anti by clan | see at GH37 | ||||||
| GH65 | L | (α/α)6 | alpha | inverting | predicted syn by clan | see at GH15 | ||||||
| GH67 | none | (β/α)8 | alpha | inverting | syn | 1gql | α-glucuronidase | Cellvibrio japonicus Ueda107 | d-glucuronic acid | Glu292 | unknown | [50] |
| GH68 | J | 5-fold β-propeller | beta | retaining | anti | 1pt2 | levansucrase | Bacillus subtilis | sucrose | Glu342 | Asp86 | [51] |
| GH70 | H | (β/α)8 | alpha | retaining | predicted anti by clan | see e.g. at GH13 | ||||||
| GH72 | A | (β/α)8 | beta | retaining | anti | 2w62 | β-1,3-glucano- transferase | Saccharomyces cerevisiae S288C | laminaripentaose | Glu176 | Glu275 | [52] |
| GH74 | none | 7-fold β-propeller | beta | inverting | syn | 2ebs | cellobiohydrolase (OXG-RCBH) | Geotrichum sp. m128 | xyloglucan heptasaccharide | Asp465 | Asp35 | [53] |
| GH77 | H | (β/α)8 | alpha | retaining | anti | 1esw | amylomaltase | Thermus aquaticus | acarbose | Asp395 | Asp293 | [54] |
| GH79 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH80 | I | α + β | beta | inverting | predicted syn by clan | see at GH24 | ||||||
| GH83 | E | 6-fold β-propeller | alpha | retaining | predicted anti by clan | see e.g. at GH33 | ||||||
| GH84 | none | (β/α)8 | beta | retaining | anti | 2chn | β-N-acetyl- glucosaminidase | Bacteroides thetaiota- omicron VPI-5482 | NAG-thiazoline | Glu242 | internal | [55] |
| GH85 | K | (β/α)8 | beta | retaining | anti | 2w92 | endo-β-N-acetyl- glucosaminidase D | Streptococcus pneumoniae TIGR4 | NAG-thiazoline | Glu337 | internal | [56] |
| GH86 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 | ||||||
| GH89 | none | (β/α)8 | alpha | retaining | anti | 2vcb | α-N-acetyl- glucosaminidase | Clostridium perfringens | PUGNAc | Glu483 | Glu601 | [57] |
| GH92 | none | (α/α)6 + β-sandw. | alpha | inverting | anti | 2ww1 | α-1,2-mannosidase | Bacteroides thetaiota- omicron VPI-5482 | thiomannobioside | Glu533 | Asp644 Asp642 | [58] |
| GH93 | E | 6-fold β-propeller | alpha | retaining | predicted anti by clan | see e.g. at GH33 | ||||||
| GH94 | none | (α/α)6 | beta | inverting | syn | 1v7x | chitobiose phosphorylase | Vibrio proteolyticus | GlcNAc | Asp492 | phosphate | [59] |
| GH95 | none | (α/α)6 | alpha | inverting | anti | 2ead | α-1,2-l-fucosidase | Bifidobacterium bifidum | Fuc-α-1,2-Gal | Glu566 | Asn423 Asp766 | [60] |
| GH97 | none | (β/α)8 | alpha | retaining + inverting | anti | 2zq0 | α-glucosidase | Bacteroides thetaiota- omicron VPI-5482 | acarbose | Glu532 | Glu508 | [61] |
| GH102 | none | double-ψ β-barrel | beta | retaining | syn | 2pi8 | lytic transglycosylase A | Escherechia coli | chitohexaose | Asp308 | none | [62] |
| GH113 | A | (β/α)8 | beta | retaining | predicted anti by clan | see e.g. at GH1 |
References
-
Heightman, T.D. and Vasella, A.T. (1999) Recent Insights into Inhibition, Structure, and Mechanism of Configuration-Retaining Glycosidases. Angewandte Chemie-International Edition 38(6), 750-770. Article online.
- Nerinckx W, Desmet T, Piens K, and Claeyssens M. (2005). An elaboration on the syn-anti proton donor concept of glycoside hydrolases: electrostatic stabilisation of the transition state as a general strategy. FEBS Lett. 2005;579(2):302-12. DOI:10.1016/j.febslet.2004.12.021 |
- Wu M, Nerinckx W, Piens K, Ishida T, Hansson H, Sandgren M, and Ståhlberg J. (2013). Rational design, synthesis, evaluation and enzyme-substrate structures of improved fluorogenic substrates for family 6 glycoside hydrolases. FEBS J. 2013;280(1):184-98. DOI:10.1111/febs.12060 |
-
Pérez S and Marchessault RH (1978) The exo-anomeric effect: experimental evidence from crystal structures. Carbohydr res 65, 114-120.
-
Cramer CJ, Truhlar DG and French AD (1997) Exo-anomeric effects on energies and geometries of different conformations of glucose and related systems in the gas phase and aqueous solution. Carbohydr res 298, 1-14.
-
pmid= 19733839
- Gloster TM, Roberts S, Perugino G, Rossi M, Moracci M, Panday N, Terinek M, Vasella A, and Davies GJ. (2006). Structural, kinetic, and thermodynamic analysis of glucoimidazole-derived glycosidase inhibitors. Biochemistry. 2006;45(39):11879-84. DOI:10.1021/bi060973x |
- van Bueren AL, Ghinet MG, Gregg K, Fleury A, Brzezinski R, and Boraston AB. (2009). The structural basis of substrate recognition in an exo-beta-D-glucosaminidase involved in chitosan hydrolysis. J Mol Biol. 2009;385(1):131-9. DOI:10.1016/j.jmb.2008.10.031 |
- Hrmova M, Varghese JN, De Gori R, Smith BJ, Driguez H, and Fincher GB. (2001). Catalytic mechanisms and reaction intermediates along the hydrolytic pathway of a plant beta-D-glucan glucohydrolase. Structure. 2001;9(11):1005-16. DOI:10.1016/s0969-2126(01)00673-6 |
- Varrot A and Davies GJ. (2003). Direct experimental observation of the hydrogen-bonding network of a glycosidase along its reaction coordinate revealed by atomic resolution analyses of endoglucanase Cel5A. Acta Crystallogr D Biol Crystallogr. 2003;59(Pt 3):447-52. DOI:10.1107/s0907444902023405 |
- Zou Jy, Kleywegt GJ, Ståhlberg J, Driguez H, Nerinckx W, Claeyssens M, Koivula A, Teeri TT, and Jones TA. (1999). Crystallographic evidence for substrate ring distortion and protein conformational changes during catalysis in cellobiohydrolase Ce16A from trichoderma reesei. Structure. 1999;7(9):1035-45. DOI:10.1016/s0969-2126(99)80171-3 |
- Sulzenbacher G, Mackenzie LF, Wilson KS, Withers SG, Dupont C, and Davies GJ. (1999). The crystal structure of a 2-fluorocellotriosyl complex of the Streptomyces lividans endoglucanase CelB2 at 1.2 A resolution. Biochemistry. 1999;38(15):4826-33. DOI:10.1021/bi982648i |
- Guérin DM, Lascombe MB, Costabel M, Souchon H, Lamzin V, Béguin P, and Alzari PM. (2002). Atomic (0.94 A) resolution structure of an inverting glycosidase in complex with substrate. J Mol Biol. 2002;316(5):1061-9. DOI:10.1006/jmbi.2001.5404 |
- Schubot FD, Kataeva IA, Chang J, Shah AK, Ljungdahl LG, Rose JP, and Wang BC. (2004). Structural basis for the exocellulase activity of the cellobiohydrolase CbhA from Clostridium thermocellum. Biochemistry. 2004;43(5):1163-70. DOI:10.1021/bi030202i |
- Suzuki R, Fujimoto Z, Ito S, Kawahara S, Kaneko S, Taira K, Hasegawa T, and Kuno A. (2009). Crystallographic snapshots of an entire reaction cycle for a retaining xylanase from Streptomyces olivaceoviridis E-86. J Biochem. 2009;146(1):61-70. DOI:10.1093/jb/mvp047 |
- Sidhu G, Withers SG, Nguyen NT, McIntosh LP, Ziser L, and Brayer GD. (1999). Sugar ring distortion in the glycosyl-enzyme intermediate of a family G/11 xylanase. Biochemistry. 1999;38(17):5346-54. DOI:10.1021/bi982946f |
- Sandgren M, Berglund GI, Shaw A, Ståhlberg J, Kenne L, Desmet T, and Mitchinson C. (2004). Crystal complex structures reveal how substrate is bound in the -4 to the +2 binding sites of Humicola grisea Cel12A. J Mol Biol. 2004;342(5):1505-17. DOI:10.1016/j.jmb.2004.07.098 |
- Uitdehaag JC, Mosi R, Kalk KH, van der Veen BA, Dijkhuizen L, Withers SG, and Dijkstra BW. (1999). X-ray structures along the reaction pathway of cyclodextrin glycosyltransferase elucidate catalysis in the alpha-amylase family. Nat Struct Biol. 1999;6(5):432-6. DOI:10.1038/8235 |
- Miyake H, Kurisu G, Kusunoki M, Nishimura S, Kitamura S, and Nitta Y. (2003). Crystal structure of a catalytic site mutant of beta-amylase from Bacillus cereus var. mycoides cocrystallized with maltopentaose. Biochemistry. 2003;42(19):5574-81. DOI:10.1021/bi020712x |
- Aleshin AE, Stoffer B, Firsov LM, Svensson B, and Honzatko RB. (1996). Crystallographic complexes of glucoamylase with maltooligosaccharide analogs: relationship of stereochemical distortions at the nonreducing end to the catalytic mechanism. Biochemistry. 1996;35(25):8319-28. DOI:10.1021/bi960321g |
- Allouch J, Helbert W, Henrissat B, and Czjzek M. (2004). Parallel substrate binding sites in a beta-agarase suggest a novel mode of action on double-helical agarose. Structure. 2004;12(4):623-32. DOI:10.1016/j.str.2004.02.020 |
- Papanikolau Y, Prag G, Tavlas G, Vorgias CE, Oppenheim AB, and Petratos K. (2001). High resolution structural analyses of mutant chitinase A complexes with substrates provide new insight into the mechanism of catalysis. Biochemistry. 2001;40(38):11338-43. DOI:10.1021/bi010505h |
- Prag G, Papanikolau Y, Tavlas G, Vorgias CE, Petratos K, and Oppenheim AB. (2000). Structures of chitobiase mutants complexed with the substrate Di-N-acetyl-d-glucosamine: the catalytic role of the conserved acidic pair, aspartate 539 and glutamate 540. J Mol Biol. 2000;300(3):611-7. DOI:10.1006/jmbi.2000.3906 |
- Vocadlo DJ, Davies GJ, Laine R, and Withers SG. (2001). Catalysis by hen egg-white lysozyme proceeds via a covalent intermediate. Nature. 2001;412(6849):835-8. DOI:10.1038/35090602 |
- Karlsen S, Hough E, Rao ZH, and Isaacs NW. (1996). Structure of a bulgecin-inhibited g-type lysozyme from the egg white of the Australian black swan. A comparison of the binding of bulgecin to three muramidases. Acta Crystallogr D Biol Crystallogr. 1996;52(Pt 1):105-14. DOI:10.1107/S0907444995008468 |
- Baldwin EP, Hajiseyedjavadi O, Baase WA, and Matthews BW. (1993). The role of backbone flexibility in the accommodation of variants that repack the core of T4 lysozyme. Science. 1993;262(5140):1715-8. DOI:10.1126/science.8259514 |
- Ducros VM, Zechel DL, Murshudov GN, Gilbert HJ, Szabó L, Stoll D, Withers SG, and Davies GJ. (2002). Substrate distortion by a beta-mannanase: snapshots of the Michaelis and covalent-intermediate complexes suggest a B(2,5) conformation for the transition state. Angew Chem Int Ed Engl. 2002;41(15):2824-7. DOI:10.1002/1521-3773(20020802)41:15<2824::AID-ANIE2824>3.0.CO;2-G |
- Garman SC, Hannick L, Zhu A, and Garboczi DN. (2002). The 1.9 A structure of alpha-N-acetylgalactosaminidase: molecular basis of glycosidase deficiency diseases. Structure. 2002;10(3):425-34. DOI:10.1016/s0969-2126(02)00726-8 |
- Abbott DW and Boraston AB. (2007). The structural basis for exopolygalacturonase activity in a family 28 glycoside hydrolase. J Mol Biol. 2007;368(5):1215-22. DOI:10.1016/j.jmb.2007.02.083 |
- Sulzenbacher G, Bignon C, Nishimura T, Tarling CA, Withers SG, Henrissat B, and Bourne Y. (2004). Crystal structure of Thermotoga maritima alpha-L-fucosidase. Insights into the catalytic mechanism and the molecular basis for fucosidosis. J Biol Chem. 2004;279(13):13119-28. DOI:10.1074/jbc.M313783200 |
- Brumshtein B, Greenblatt HM, Butters TD, Shaaltiel Y, Aviezer D, Silman I, Futerman AH, and Sussman JL. (2007). Crystal structures of complexes of N-butyl- and N-nonyl-deoxynojirimycin bound to acid beta-glucosidase: insights into the mechanism of chemical chaperone action in Gaucher disease. J Biol Chem. 2007;282(39):29052-29058. DOI:10.1074/jbc.M705005200 |
- Sim L, Quezada-Calvillo R, Sterchi EE, Nichols BL, and Rose DR. (2008). Human intestinal maltase-glucoamylase: crystal structure of the N-terminal catalytic subunit and basis of inhibition and substrate specificity. J Mol Biol. 2008;375(3):782-92. DOI:10.1016/j.jmb.2007.10.069 |
- Verhaest M, Lammens W, Le Roy K, De Ranter CJ, Van Laere A, Rabijns A, and Van den Ende W. (2007). Insights into the fine architecture of the active site of chicory fructan 1-exohydrolase: 1-kestose as substrate vs sucrose as inhibitor. New Phytol. 2007;174(1):90-100. DOI:10.1111/j.1469-8137.2007.01988.x |
- Amaya MF, Watts AG, Damager I, Wehenkel A, Nguyen T, Buschiazzo A, Paris G, Frasch AC, Withers SG, and Alzari PM. (2004). Structural insights into the catalytic mechanism of Trypanosoma cruzi trans-sialidase. Structure. 2004;12(5):775-84. DOI:10.1016/j.str.2004.02.036 |
- Varghese JN, McKimm-Breschkin JL, Caldwell JB, Kortt AA, and Colman PM. (1992). The structure of the complex between influenza virus neuraminidase and sialic acid, the viral receptor. Proteins. 1992;14(3):327-32. DOI:10.1002/prot.340140302 |
- Rojas AL, Nagem RA, Neustroev KN, Arand M, Adamska M, Eneyskaya EV, Kulminskaya AA, Garratt RC, Golubev AM, and Polikarpov I. (2004). Crystal structures of beta-galactosidase from Penicillium sp. and its complex with galactose. J Mol Biol. 2004;343(5):1281-92. DOI:10.1016/j.jmb.2004.09.012 |
- Gibson RP, Gloster TM, Roberts S, Warren RA, Storch de Gracia I, García A, Chiara JL, and Davies GJ. (2007). Molecular basis for trehalase inhibition revealed by the structure of trehalase in complex with potent inhibitors. Angew Chem Int Ed Engl. 2007;46(22):4115-9. DOI:10.1002/anie.200604825 |
- Numao S, Kuntz DA, Withers SG, and Rose DR. (2003). Insights into the mechanism of Drosophila melanogaster Golgi alpha-mannosidase II through the structural analysis of covalent reaction intermediates. J Biol Chem. 2003;278(48):48074-83. DOI:10.1074/jbc.M309249200 |
- Yang JK, Yoon HJ, Ahn HJ, Lee BI, Pedelacq JD, Liong EC, Berendzen J, Laivenieks M, Vieille C, Zeikus GJ, Vocadlo DJ, Withers SG, and Suh SW. (2004). Crystal structure of beta-D-xylosidase from Thermoanaerobacterium saccharolyticum, a family 39 glycoside hydrolase. J Mol Biol. 2004;335(1):155-65. DOI:10.1016/j.jmb.2003.10.026 |
- Hidaka M, Fushinobu S, Ohtsu N, Motoshima H, Matsuzawa H, Shoun H, and Wakagi T. (2002). Trimeric crystal structure of the glycoside hydrolase family 42 beta-galactosidase from Thermus thermophilus A4 and the structure of its complex with galactose. J Mol Biol. 2002;322(1):79-91. DOI:10.1016/s0022-2836(02)00746-5 |
- Kitago Y, Karita S, Watanabe N, Kamiya M, Aizawa T, Sakka K, and Tanaka I. (2007). Crystal structure of Cel44A, a glycoside hydrolase family 44 endoglucanase from Clostridium thermocellum. J Biol Chem. 2007;282(49):35703-11. DOI:10.1074/jbc.M706835200 |
- Davies GJ, Dodson G, Moore MH, Tolley SP, Dauter Z, Wilson KS, Rasmussen G, and Schülein M. (1996). Structure determination and refinement of the Humicola insolens endoglucanase V at 1.5 A resolution. Acta Crystallogr D Biol Crystallogr. 1996;52(Pt 1):7-17. DOI:10.1107/S0907444995009280 |
- Karaveg K, Siriwardena A, Tempel W, Liu ZJ, Glushka J, Wang BC, and Moremen KW. (2005). Mechanism of class 1 (glycosylhydrolase family 47) {alpha}-mannosidases involved in N-glycan processing and endoplasmic reticulum quality control. J Biol Chem. 2005;280(16):16197-207. DOI:10.1074/jbc.M500119200 |
- Cantú D, Nerinckx W, and Reilly PJ. (2008). Theory and computation show that Asp463 is the catalytic proton donor in human endoplasmic reticulum alpha-(1-->2)-mannosidase I. Carbohydr Res. 2008;343(13):2235-42. DOI:10.1016/j.carres.2008.05.026 |
- Hövel K, Shallom D, Niefind K, Belakhov V, Shoham G, Baasov T, Shoham Y, and Schomburg D. (2003). Crystal structure and snapshots along the reaction pathway of a family 51 alpha-L-arabinofuranosidase. EMBO J. 2003;22(19):4922-32. DOI:10.1093/emboj/cdg494 |
- Miyanaga A, Koseki T, Matsuzawa H, Wakagi T, Shoun H, and Fushinobu S. (2004). Crystal structure of a family 54 alpha-L-arabinofuranosidase reveals a novel carbohydrate-binding module that can bind arabinose. J Biol Chem. 2004;279(43):44907-14. DOI:10.1074/jbc.M405390200 |
- Ishida T, Fushinobu S, Kawai R, Kitaoka M, Igarashi K, and Samejima M. (2009). Crystal structure of glycoside hydrolase family 55 {beta}-1,3-glucanase from the basidiomycete Phanerochaete chrysosporium. J Biol Chem. 2009;284(15):10100-9. DOI:10.1074/jbc.M808122200 |
-
Marković-Housley Z, Miglierini G, Soldatova L, Rizkallah PJ, Müller U, Schirmer T. Crystal structure of hyaluronidase, a major allergen of bee venom. Structure. 2000 Oct 15;8(10):1025-35.
Note: Due to a problem with PubMed data, this reference is not automatically formatted. Please see these links out: DOI:10.1016/S0969-2126(00)00511-6 PMID:11080624
- Imamura H, Fushinobu S, Yamamoto M, Kumasaka T, Jeon BS, Wakagi T, and Matsuzawa H. (2003). Crystal structures of 4-alpha-glucanotransferase from Thermococcus litoralis and its complex with an inhibitor. J Biol Chem. 2003;278(21):19378-86. DOI:10.1074/jbc.M213134200 |
- Nurizzo D, Nagy T, Gilbert HJ, and Davies GJ. (2002). The structural basis for catalysis and specificity of the Pseudomonas cellulosa alpha-glucuronidase, GlcA67A. Structure. 2002;10(4):547-56. DOI:10.1016/s0969-2126(02)00742-6 |
- Meng G and Fütterer K. (2003). Structural framework of fructosyl transfer in Bacillus subtilis levansucrase. Nat Struct Biol. 2003;10(11):935-41. DOI:10.1038/nsb974 |
- Hurtado-Guerrero R, Schüttelkopf AW, Mouyna I, Ibrahim AF, Shepherd S, Fontaine T, Latgé JP, and van Aalten DM. (2009). Molecular mechanisms of yeast cell wall glucan remodeling. J Biol Chem. 2009;284(13):8461-9. DOI:10.1074/jbc.M807990200 |
- Yaoi K, Kondo H, Hiyoshi A, Noro N, Sugimoto H, Tsuda S, Mitsuishi Y, and Miyazaki K. (2007). The structural basis for the exo-mode of action in GH74 oligoxyloglucan reducing end-specific cellobiohydrolase. J Mol Biol. 2007;370(1):53-62. DOI:10.1016/j.jmb.2007.04.035 |
- Przylas I, Terada Y, Fujii K, Takaha T, Saenger W, and Sträter N. (2000). X-ray structure of acarbose bound to amylomaltase from Thermus aquaticus. Implications for the synthesis of large cyclic glucans. Eur J Biochem. 2000;267(23):6903-13. DOI:10.1046/j.1432-1033.2000.01790.x |
- Dennis RJ, Taylor EJ, Macauley MS, Stubbs KA, Turkenburg JP, Hart SJ, Black GN, Vocadlo DJ, and Davies GJ. (2006). Structure and mechanism of a bacterial beta-glucosaminidase having O-GlcNAcase activity. Nat Struct Mol Biol. 2006;13(4):365-71. DOI:10.1038/nsmb1079 |
- Abbott DW, Macauley MS, Vocadlo DJ, and Boraston AB. (2009). Streptococcus pneumoniae endohexosaminidase D, structural and mechanistic insight into substrate-assisted catalysis in family 85 glycoside hydrolases. J Biol Chem. 2009;284(17):11676-89. DOI:10.1074/jbc.M809663200 |
- Ficko-Blean E, Stubbs KA, Nemirovsky O, Vocadlo DJ, and Boraston AB. (2008). Structural and mechanistic insight into the basis of mucopolysaccharidosis IIIB. Proc Natl Acad Sci U S A. 2008;105(18):6560-5. DOI:10.1073/pnas.0711491105 |
-
Zhu et al. (2010) Nature Chemical Biology in the press; DOI: 10.1038/nchembio.278 direct link.
- Hidaka M, Honda Y, Kitaoka M, Nirasawa S, Hayashi K, Wakagi T, Shoun H, and Fushinobu S. (2004). Chitobiose phosphorylase from Vibrio proteolyticus, a member of glycosyl transferase family 36, has a clan GH-L-like (alpha/alpha)(6) barrel fold. Structure. 2004;12(6):937-47. DOI:10.1016/j.str.2004.03.027 |
- Nagae M, Tsuchiya A, Katayama T, Yamamoto K, Wakatsuki S, and Kato R. (2007). Structural basis of the catalytic reaction mechanism of novel 1,2-alpha-L-fucosidase from Bifidobacterium bifidum. J Biol Chem. 2007;282(25):18497-18509. DOI:10.1074/jbc.M702246200 |
- Kitamura M, Okuyama M, Tanzawa F, Mori H, Kitago Y, Watanabe N, Kimura A, Tanaka I, and Yao M. (2008). Structural and functional analysis of a glycoside hydrolase family 97 enzyme from Bacteroides thetaiotaomicron. J Biol Chem. 2008;283(52):36328-37. DOI:10.1074/jbc.M806115200 |
- van Straaten KE, Barends TR, Dijkstra BW, and Thunnissen AM. (2007). Structure of Escherichia coli Lytic transglycosylase MltA with bound chitohexaose: implications for peptidoglycan binding and cleavage. J Biol Chem. 2007;282(29):21197-205. DOI:10.1074/jbc.M701818200 |